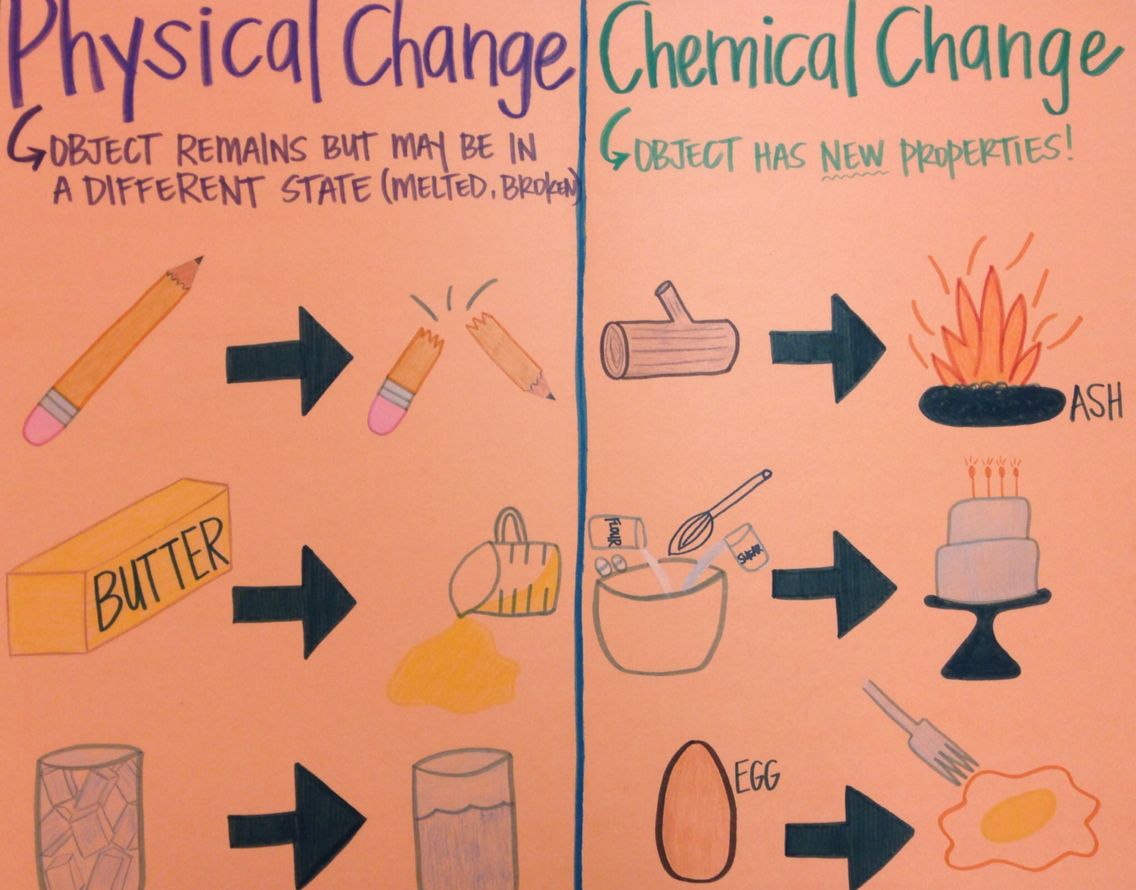
For example, the reaction between salt and water, or the dissolution of salt in water, involves breaking the bonds between ions, but it also creates ion-dipole interactions between the water and ions. However, some reactions can go either way and sometimes physical processes involve the breaking of chemical bonds. Since physical changes are only a transformation that changes a substance's properties without undergoing a chemical change, they are reversible. Conversely, the paper’s molecules keep the same atomic structure, but the interaction between the paper molecules is altered, or broken. The ice’s molecules maintain the same atomic structure (H2O), but more hydrogen bonds between each water molecule are formed. Some examples are freezing water and cutting paper. Physical changes are usually intermolecular changes (literally meaning “between molecules”), such as phase changes. Rusting: The chemical reaction that occurs when iron reacts with water and oxygen to form iron oxide (rust).ĭigestion: The chemical reactions that occur in the body to break down food into nutrients that can be absorbed and used by cells. Examples of chemical changes include:īurning: The chemical reaction that occurs when a substance reacts with oxygen to produce heat and light. Remember that chemical changes are usually accompanied by a chemical reaction to show this breaking and forming of bonds. They are rather expressed with brackets and charges around the brackets to show the transfer of electrons. Note that ionic bonds are not represented like covalent bonds with lines as shown in the above diagram. Here, we can see that the oxygen molecule's covalent bonds had to break, and then new ionic bonds formed between the magnesium and oxygen to form two molecules of MgO. The bonds that are broken are red, and the bonds formed are green. The synthesis reaction above shows an interaction between two magnesium atoms and an oxygen molecule. Tearing Paper Crystallization of Sugar The crystallization of sugar is indeed a classic example of a physical change.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
